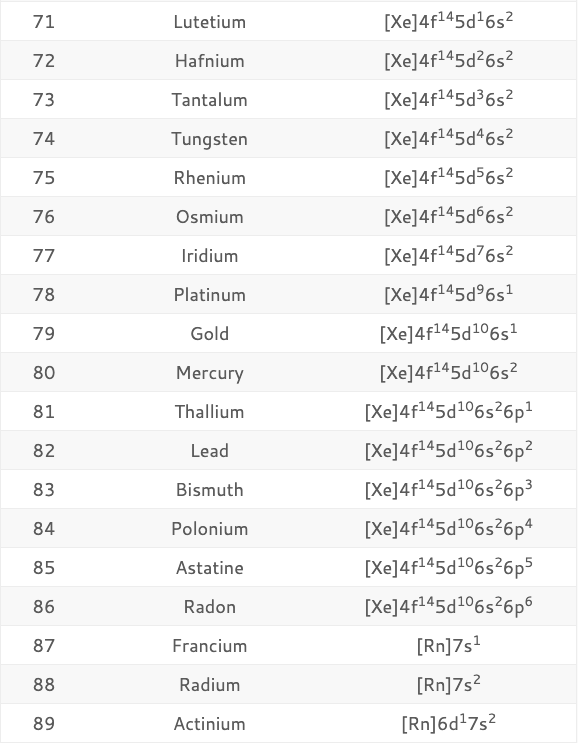
Therefore, the number of electrons in an atom of an element is equal to the element's atomic number (Z): The number of protons in the nucleus of an atom is equal to its atomic number (Z). That is, for an atom to be neutral (no overall charge), the number of negative charges (electrons) must be equal to the number of positive charges (protons). The number of electrons around the nucleus of each atom is equal to the number of protons in its nucleus. No ads = no money for us = no free stuff for you! Electron Shell Concept
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
